Nurotron products are available in 20+ countries. 20000+ users from worldwide have felt the life changes with Nurotron products.

Founded in 2006, headquarter in Hangzhou, China and R&D center in Califonia U.S., Nurotron is dedicated to the rehabilitation of people with disabilities.
Nurotron manufactures neural electrical stimulation therapy and rehabilitation products including:
Product Application Fields: The WHO estimates that about 5% of the world’s population suffers from disabling hearing loss, affecting their health and quality of life.
Nurotron products are available in 20+ countries.
20000+ users from worldwide have felt the life changes with Nurotron products.
To be a world leading medical equipment device group.
Help the hearing impaired hear. Help the visually impaired see. Help the paralytic walk.
In 2006, Nurotron R&D center in the U.S. was established







Our company was established

We completed the clinical trial surgeries for the Cochlear Implant Systems 1.0
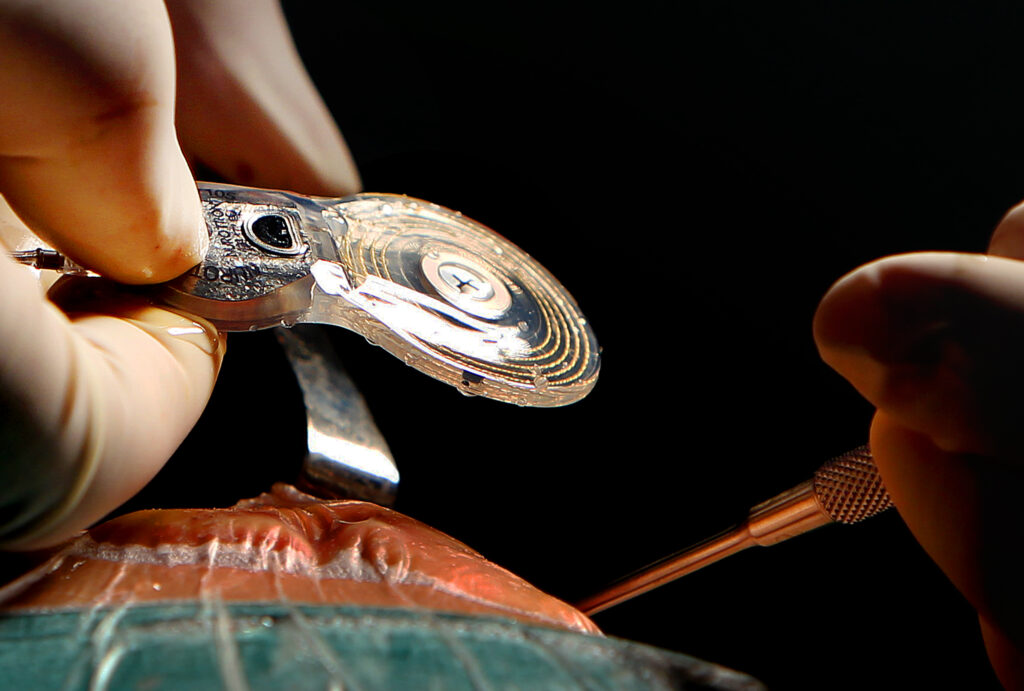
Our NCIS 1.0 product, including its two components, the implant 1.0 ("CS-10A") and the speech processor 1.0 ("Venus") obtained the NMPA approval for use in patients aged 6 years and older

Our NCIS 1.0 product, including its two components, the implant 1.0 ("CS-10A") and the speech processor 1.0 ("Venus") obtained the NMPA approval for use in patients aged 1 year and older

Our company received the second-class National Scientific and Technological Progress Award

Our speech processor 2.0 ("Enduro"), a component of our NCIS2.0 product obtained the NMPA approval

Our implant 2.0 ("CS-20A") and speech processor 3.0("Voyager") obtained the NMPA approval

Our company was awarded China National Specialized, Sophisticated, Unique and Innovative “Little Giant” Enterprise

Our company received the second-class National Scientific and Technological Progress Award

Our implant 3.0 (“CS-30A”) obtained the NMPA approval